 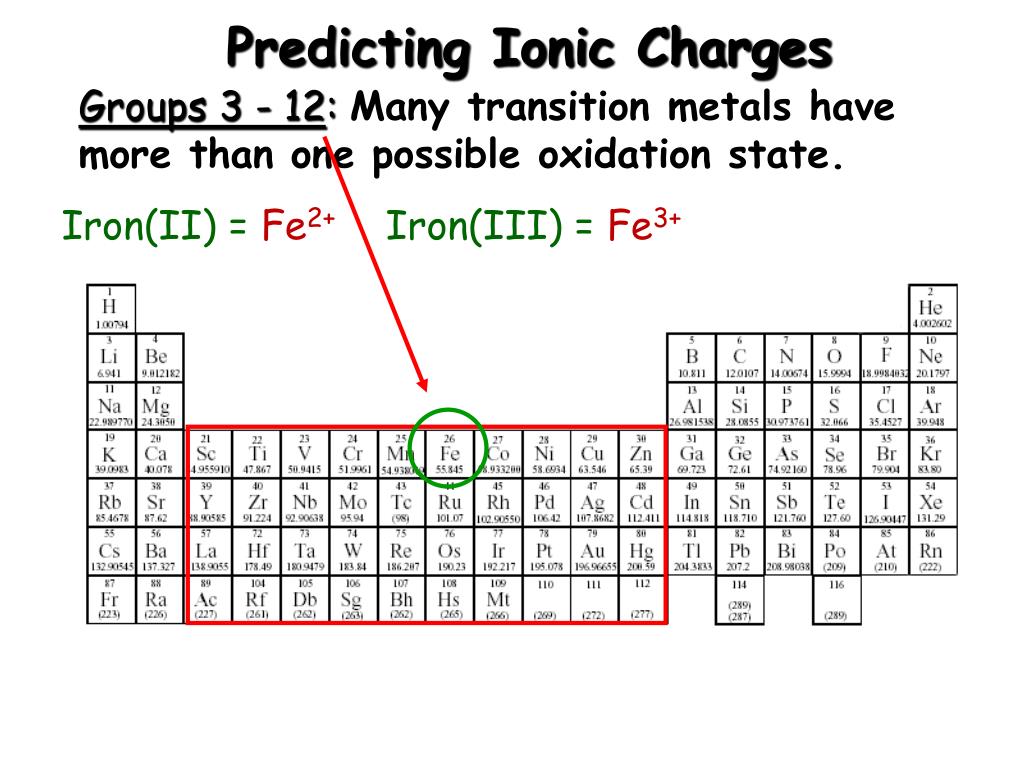
(Remember from our conventions for writing formulas that we do not write a 1 subscript if there is only one atom of a particular element present. The formula Na 2Cl 2 also has balanced charges, but the convention is to use the lowest ratio of ions, which would be one of each. The Roman numeral IV gives +4 as tin's charge.\)). Look at Least-Common-Multiple.html if you're not sure about how the subscripts in iron(III) sulfide came to be.Įxample #4 - Write the formula for: tin(IV) phosphide Step #4 - since a formula must have zero total charge, you write the formula Fe 2S 3. Step #3 - Sulfide (the anion) means S 2¯. remember, that comes from the Roman numeral. Step #2 - the charge on the cation is a positive three. Iron(II) sulfide Iron(II) sulfide Other names Iron sulfideIron sulphide Identifiers CAS number 1 Properties Molecular formula FeS Molar mass. Step #1 - the symbol of the cation is Fe. Step #4 - since a formula must have zero total charge, you write the formula Cu 2O.Įxample #3 - Write the formula for: iron(III) sulfide 
Step #3 - the anion symbol and charge comes from the second name. Step #2 - the Roman numeral WILL tell you the charge on the cation. 
Step #1 - the first word tells you the symbol of the cation. Chloride is the name and Cl¯ is the face.Įxample #2 - Write the formula for: copper(I) oxide The ChemTeam is often asked by students, "But how do you know that chloride means Cl¯?" That type of question is usually answered with a question, as in "How do you know the name and face of your best friend?" That's right, you've spent time in their company, to the point where you have memorized the connection between name and face. On writing the formula from the ions, you may want to review Charge-Crossing.html or Least-Common-Multiple.html for more information. Step #4 - remembering the rule that a formula must have zero total charge, you write the formula CuCl 2. This suggestion has not been followed, but the Stock system remains in use world-wide.Įxample #1 - Write the formula for: copper(II) chloride In 1934, Stock approved of the Roman numerals, but felt it better to keep the hyphen and drop the parenthesis. For example, FeCl 2,which would have been named iron(2)-chloride according to Stock's original idea, became iron(II) chloride in the revised proposal. In 1924, a German commission recommended Stock's system be adopted with some changes. In his own words, he considered the system to be "simple, clear, immediately intelligible, capable of the most general application." Look at Least-Common-Multiple.html if youre not sure about how the subscripts in iron(III) sulfide came to be. Step 4 - since a formula must have zero total charge, you write the formula Fe 2 S 3. Step 3 - Sulfide (the anion) means S 2 ¯. It was designed by Alfred Stock (1876-1946), a German chemist and first published in 1919. Step 2 - the charge on the cation is a positive three. The type of naming you will learn about is called the Stock system or Stock's system. The ChemTeam holds their students responsible for: Cu, Fe, Hg, Pb, Sn, Mn, Co, Au, and Cr. Your teacher will hold you responsible for the cations you must learn. Leave out all charges and all subscripts that are 1. 
The anions involved have only one charge. Iron(III) sulfide - Wikipedia Writing symbol equations - BBC Bitesize Writing word equations - BBC. The cations involved in this lesson have AT LEAST TWO charges. The four formulas above are all examples of this type. This lesson shows you how to write the formula of a binary compound from the word name when a cation of variable charge is involved. There can also be several of each element such as Fe 2O 3 or SnBr 4. There can be one of each element such as in CuCl or FeO. ChemTeam: Nomenclature-Binary Compounds:Stockīinary Compounds of Cations with Variable ChargesĪ binary compound is one made of two different elements. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
